Pets can be born with heart disease (congenital) or they may develop heart issues with age. At Veterinary Specialty Center, our pet cardiology team in Bannockburn, IL, focuses on addressing diseases related to your pet’s heart and lungs. Specifically, we concentrate on diagnosing and treating congestive heart failure, damage to the heart muscle or valves, cardiac arrhythmias, congenital defects, diseases of the pericardium, high blood pressure, coughing, and other breathing issues.
Some pets with heart disease may show symptoms such as trouble breathing, coughing, lethargy, weakness, and fainting. Other conditions, such as a heart murmur, may not show outward signs of heart disease but typically require routine monitoring with or without treatment based on cause and severity.

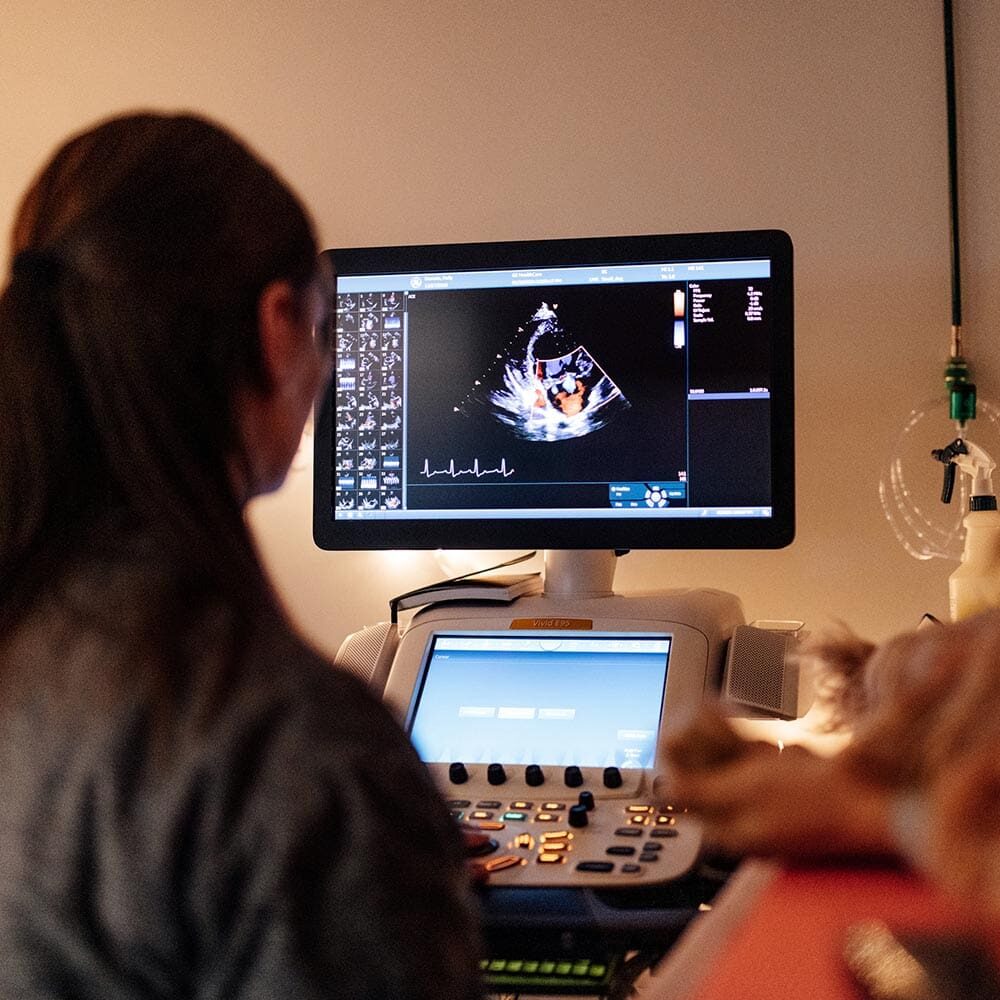
What to Expect from a Pet Cardiology Appointment
During a cardiology visit, a veterinary technician will collect your pet's medical history before your veterinarian performs a cardiovascular physical examination. Your cardiologist may conduct the following diagnostic tests based on your pet’s examination results:
-
Echocardiography (ultrasound of the heart)
-
Chest X-rays
-
Blood pressure measurement
-
Electrocardiography (ECG; heart rhythm evaluation)
-
Holter monitoring (home ECG monitors)
-
Blood work
Your cardiologist will review your pet’s results with you at the end of the appointment. If you have questions about your pet’s procedures and recommended treatments, you will have plenty of time to speak with them and get the information you need.
Cardiology appointments typically take 1-2 hours, depending on the diagnostics needed. Follow-up care may involve your primary care veterinarian or our pet cardiology team.
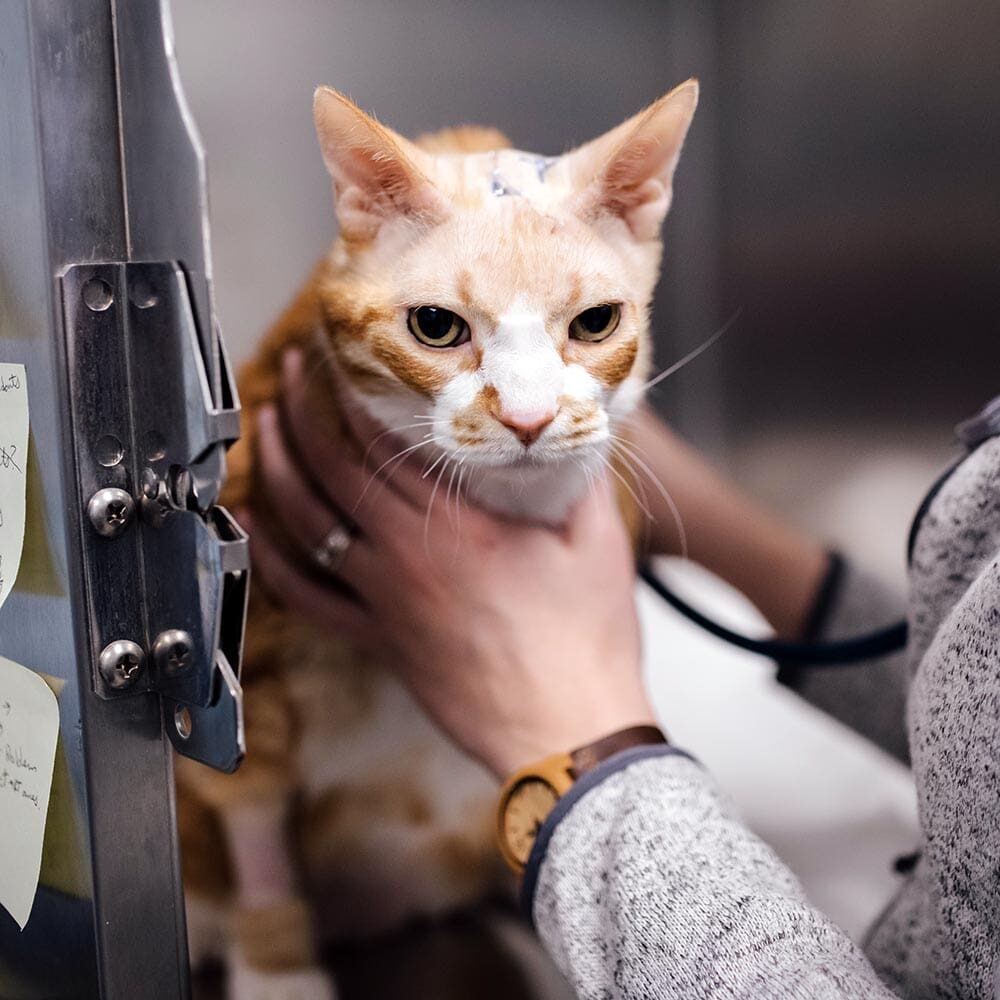
Common Cardiac Issues in Pets
Our pet cardiologists are extensively trained in recognizing and diagnosing many heart- and lung-related conditions in dogs and cats, including:
-
Dilated Cardiomyopathy (DCM)
-
Hypertrophic Cardiomyopathy (HCM)
-
Pulmonary Hypertension
Minimally Invasive Procedures
The cardiology specialists of VSC are highly experienced in performing various minimally invasive, image-guided, cardiac catheterization procedures, including pacemaker implantations, balloon valvuloplasties, and PDA occlusions with Amplatz Canine Duct Occluder (ACDO) devices.
-
Balloon Valvuloplasty for Pulmonic Stenosis
-
Patent Ductus Arteriosus Occlusion
-
Transvenous Pacemaker Implantation

